Ocean Acidification, A Global Threat To Marine Life
Introduction
Ocean acidification is sometimes called “climate change’s equally evil twin,” and for good reason: it's a significant and harmful consequence of excess carbon dioxide in the atmosphere that we don't see or feel because its effects are happening underwater. At least one-quarter of the carbon dioxide (CO2) released by burning coal, oil and gas doesn't stay in the air, but instead dissolves into the ocean. Since the beginning of the industrial era, the ocean has absorbed some 525 billion tons of CO2 from the atmosphere, presently around 22 million tons per day.
At first, scientists thought that this might be a good thing because it leaves less carbon dioxide in the air to warm the planet. But in the past decade, they’ve realized that this slowed warming has come at the cost of changing the ocean’s chemistry. When carbon dioxide dissolves in seawater, the water becomes more acidic and the ocean’s pH (a measure of how acidic or basic the ocean is) drops. Even though the ocean is immense, enough carbon dioxide can have a major impact. In the past 200 years alone, ocean water has become 30 percent more acidic—faster than any known change in ocean chemistry in the last 50 million years.
Scientists formerly didn’t worry about this process because they always assumed that rivers carried enough dissolved chemicals from rocks to the ocean to keep the ocean’s pH stable. (Scientists call this stabilizing effect “buffering.”) But so much carbon dioxide is dissolving into the ocean so quickly that this natural buffering hasn’t been able to keep up, resulting in relatively rapidly dropping pH in surface waters. As those surface layers gradually mix into deep water, the entire ocean is affected.
Such a relatively quick change in ocean chemistry doesn’t give marine life, which evolved over millions of years in an ocean with a generally stable pH, much time to adapt. In fact, the shells of some animals are already dissolving in the more acidic seawater, and that’s just one way that acidification may affect ocean life. Overall, it's expected to have dramatic and mostly negative impacts on ocean ecosystems—although some species (especially those that live in estuaries) are finding ways to adapt to the changing conditions.
However, while the chemistry is predictable, the details of the biological impacts are not. Although scientists have been tracking ocean pH for more than 30 years, biological studies really only started in 2003, when the rapid shift caught their attention and the term "ocean acidification" was first coined. What we do know is that things are going to look different, and we can't predict in any detail how they will look. Some organisms will survive or even thrive under the more acidic conditions while others will struggle to adapt, and may even go extinct. Beyond lost biodiversity, acidification will affect fisheries and aquaculture, threatening food security for millions of people, as well as tourism and other sea-related economies.
Acidification Chemistry
At its core, the issue of ocean acidification is simple chemistry. There are two important things to remember about what happens when carbon dioxide dissolves in seawater. First, the pH of seawater water gets lower as it becomes more acidic. Second, this process binds up carbonate ions and makes them less abundant—ions that corals, oysters, mussels, and many other shelled organisms need to build shells and skeletons.
A More Acidic Ocean
Carbon dioxide is naturally in the air: plants need it to grow, and animals exhale it when they breathe. But, thanks to people burning fuels, there is now more carbon dioxide in the atmosphere than anytime in the past 15 million years. Most of this CO2 collects in the atmosphere and, because it absorbs heat from the sun, creates a blanket around the planet, warming its temperature. But some 30 percent of this CO2 dissolves into seawater, where it doesn't remain as floating CO2 molecules. A series of chemical changes break down the CO2 molecules and recombine them with others.
When water (H2O) and CO2 mix, they combine to form carbonic acid (H2CO3). Carbonic acid is weak compared to some of the well-known acids that break down solids, such as hydrochloric acid (the main ingredient in gastric acid, which digests food in your stomach) and sulfuric acid (the main ingredient in car batteries, which can burn your skin with just a drop). The weaker carbonic acid may not act as quickly, but it works the same way as all acids: it releases hydrogen ions (H+), which bond with other molecules in the area.
Seawater that has more hydrogen ions is more acidic by definition, and it also has a lower pH. In fact, the definitions of acidification terms—acidity, H+, pH —are interlinked: acidity describes how many H+ ions are in a solution; an acid is a substance that releases H+ ions; and pH is the scale used to measure the concentration of H+ ions.
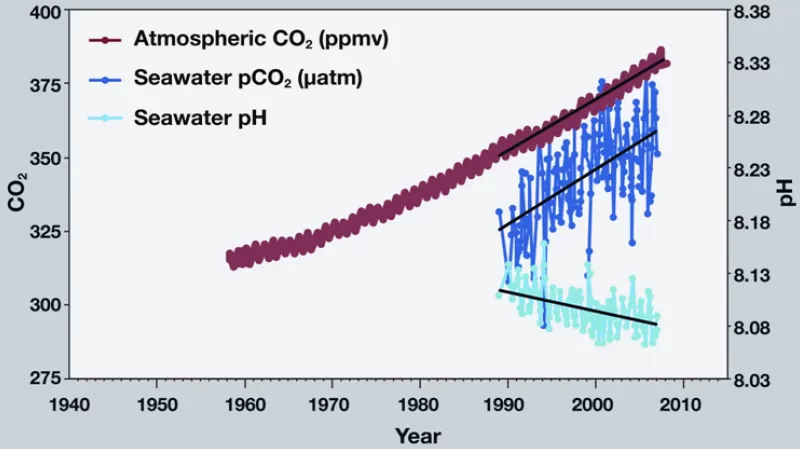
The lower the pH, the more acidic the solution. The pH scale goes from extremely basic at 14 (lye has a pH of 13) to extremely acidic at 1 (lemon juice has a pH of 2), with a pH of 7 being neutral (neither acidic or basic). The ocean itself is not actually acidic in the sense of having a pH less than 7, and it won’t become acidic even with all the CO2 that is dissolving into the ocean. But the changes in the direction of increasing acidity are still dramatic.
So far, ocean pH has dropped from 8.2 to 8.1 since the industrial revolution, and is expected by fall another 0.3 to 0.4 pH units by the end of the century. A drop in pH of 0.1 might not seem like a lot, but the pH scale, like the Richter scale for measuring earthquakes, is logarithmic. For example, pH 4 is ten times more acidic than pH 5 and 100 times (10 times 10) more acidic than pH 6. If we continue to add carbon dioxide at current rates, seawater pH may drop another 120 percent by the end of this century, to 7.8 or 7.7, creating an ocean more acidic than any seen for the past 20 million years or more.
Why Acidity Matters

Many chemical reactions, including those that are essential for life, are sensitive to small changes in pH. In humans, for example, normal blood pH ranges between 7.35 and 7.45. A drop in blood pH of 0.2-0.3 can cause seizures, comas, and even death. Similarly, a small change in the pH of seawater can have harmful effects on marine life, impacting chemical communication, reproduction, and growth.
The building of skeletons in marine creatures is particularly sensitive to acidity. One of the molecules that hydrogen ions bond with is carbonate (CO3-2), a key component of calcium carbonate (CaCO3) shells. To make calcium carbonate, shell-building marine animals such as corals and oysters combine a calcium ion (Ca+2) with carbonate (CO3-2) from surrounding seawater, releasing carbon dioxide and water in the process.
Like calcium ions, hydrogen ions tend to bond with carbonate—but they have a greater attraction to carbonate than calcium. When a hydrogen bonds with carbonate, a bicarbonate ion (HCO3-) is formed. Shell-building organisms can't extract the carbonate ion they need from bicarbonate, preventing them from using that carbonate to grow new shell. In this way, the hydrogen essentially binds up the carbonate ions, making it harder for shelled animals to build their homes. Even if animals are able to build skeletons in more acidic water, they may have to spend more energy to do so, taking away resources from other activities like reproduction. If there are too many hydrogen ions around and not enough molecules for them to bond with, they can even begin breaking existing calcium carbonate molecules apart—dissolving shells that already exist.
This is just one process that extra hydrogen ions—caused by dissolving carbon dioxide—may interfere with in the ocean. Organisms in the water, thus, have to learn to survive as the water around them has an increasing concentration of carbonate-hogging hydrogen ions.
Impacts on Ocean Life
The pH of the ocean fluctuates within limits as a result of natural processes, and ocean organisms are well-adapted to survive the changes that they normally experience. Some marine species may be able to adapt to more extreme changes—but many will suffer, and there will likely be extinctions. We can't know this for sure, but during the last great acidification event 55 million years ago, there were mass extinctions in some species including deep sea invertebrates. A more acidic ocean won’t destroy all marine life in the sea, but the rise in seawater acidity of 30 percent that we have already seen is already affecting some ocean organisms.
Coral Reefs

Reef-building corals craft their own homes from calcium carbonate, forming complex reefs that house the coral animals themselves and provide habitat for many other organisms. Acidification may limit coral growth by corroding pre-existing coral skeletons while simultaneously slowing the growth of new ones, and the weaker reefs that result will be more vulnerable to erosion. This erosion will come not only from storm waves, but also from animals that drill into or eat coral. A recent study predicts that by roughly 2080 ocean conditions will be so acidic that even otherwise healthy coral reefs will be eroding more quickly than they can rebuild.
Acidification may also impact corals before they even begin constructing their homes. The eggs and larvae of only a few coral species have been studied, and more acidic water didn’t hurt their development while they were still in the plankton. However, larvae in acidic water had more trouble finding a good place to settle, preventing them from reaching adulthood.
How much trouble corals run into will vary by species. Some types of coral can use bicarbonate instead of carbonate ions to build their skeletons, which gives them more options in an acidifying ocean. Some can survive without a skeleton and return to normal skeleton-building activities once the water returns to a more comfortable pH. Others can handle a wider pH range.
Nonetheless, in the next century we will see the common types of coral found in reefs shifting—though we can't be entirely certain what that change will look like. On reefs in Papua New Guinea that are affected by natural carbon dioxide seeps, big boulder colonies have taken over and the delicately branching forms have disappeared, probably because their thin branches are more susceptible to dissolving. This change is also likely to affect the many thousands of organisms that live among the coral, including those that people fish and eat, in unpredictable ways. In addition, acidification gets piled on top of all the other stresses that reefs have been suffering from, such as warming water (which causes another threat to reefs known as coral bleaching), pollution, and overfishing.
Looking to the Future
If the amount of carbon dioxide in the atmosphere stabilizes, eventually buffering (or neutralizing) will occur and pH will return to normal. This is why there are periods in the past with much higher levels of carbon dioxide but no evidence of ocean acidification: the rate of carbon dioxide increase was slower, so the ocean had time to buffer and adapt. But this time, pH is dropping too quickly. Buffering will take thousands of years, which is way too long a period of time for the ocean organisms affected now and in the near future.
So far, the signs of acidification visible to humans are few. But they will only increase as more carbon dioxide dissolves into seawater over time.